System for the Preparation of Autologous
Platelet-Rich Fibrin Matrix (PRFM)
About the SELPHYL® PRFM System
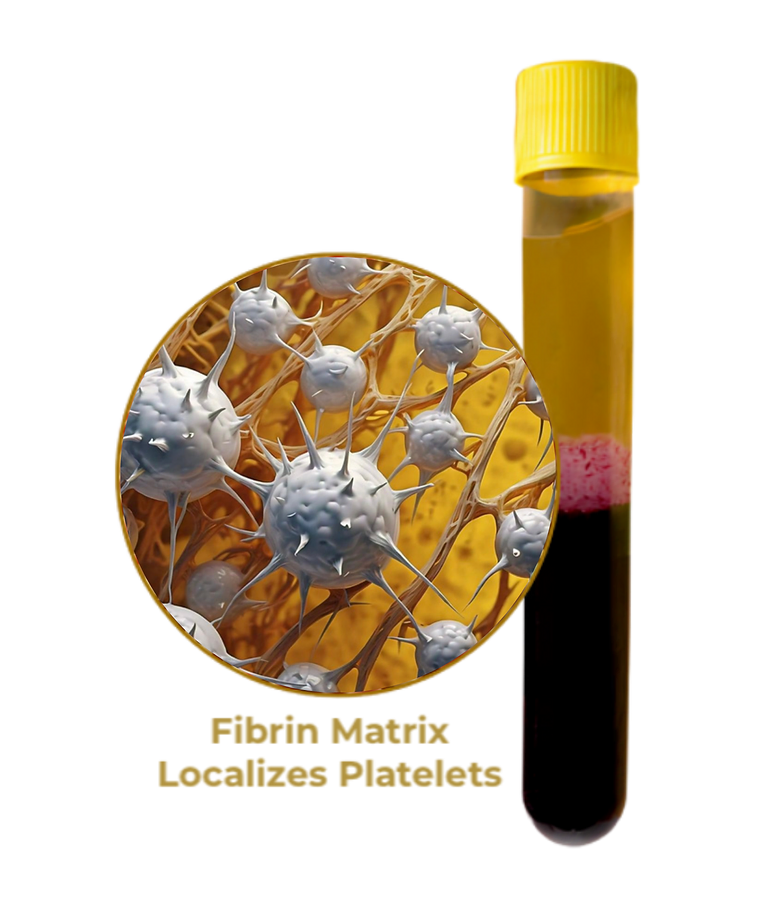
- PRFM Scaffold Protects Platelets
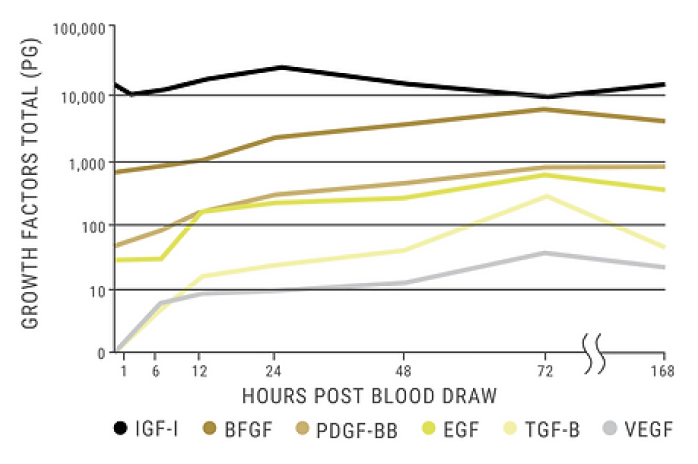
- Platelets Release Growth Factors Up to 7 Days *
- Easy to Use and Reproducible
- Full Training and Continued Support for Staff
As Seen In


See the Magic of SELPHYL® in 5 Easy Steps
The Strength of Platelets
Platelets and other blood components migrate to injury sites, releasing growth factors that initiate and promote healing. Concentrating platelets at these sites can enhance the body's natural healing capacity.
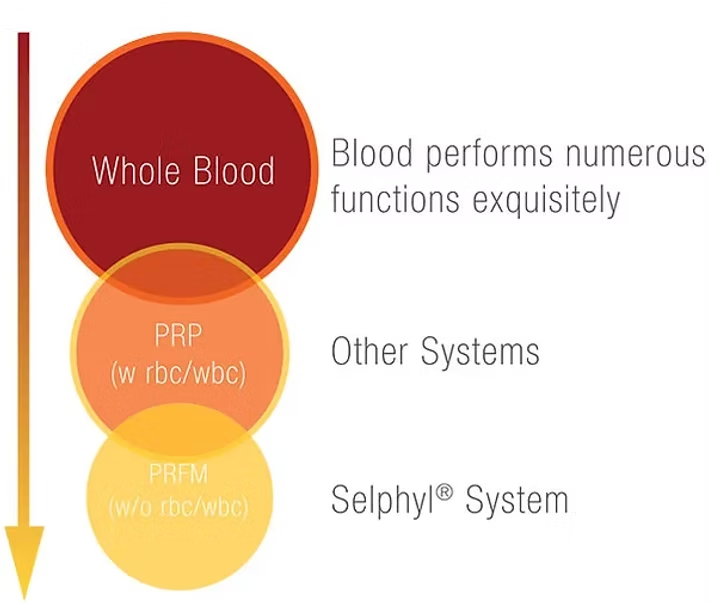
How is SELPHYL® PRFM Different from PRP?
Traditional
PRP
SELPHYL®
PRFM
Growth Factor Total (pg)

Hours Post Blood Draw
Growth Factor Total (pg)

Hours Post Blood Draw
The Science Behind SELPHYL®
Platelets Release Growth Factors
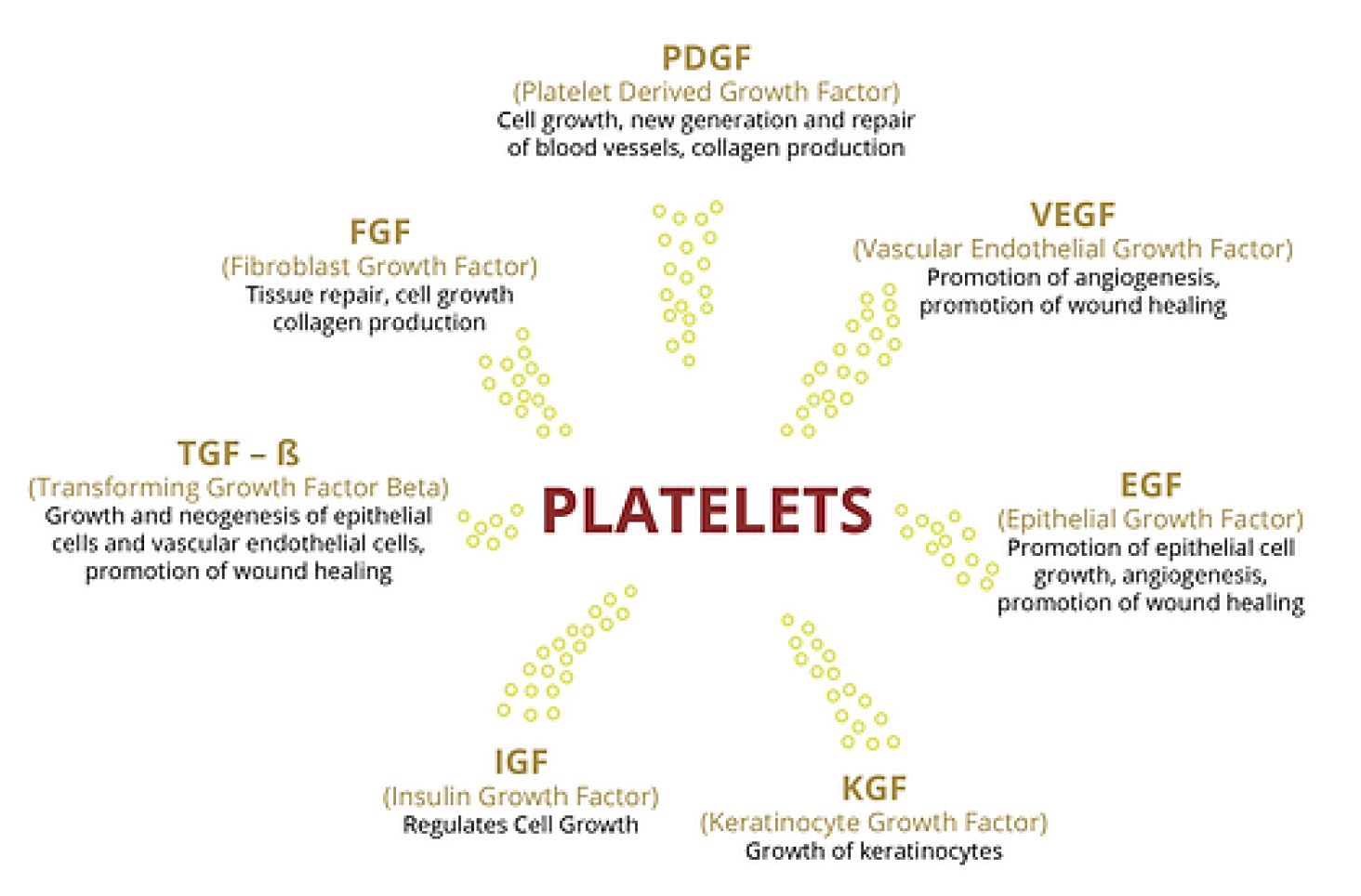
As soon as SELPHYL® - prepared PRFM is administered, growth factors are released through the formation of a three dimensional cross-linked fibrin matrix which is essential to the platelet plug as it serves as a binding site for both platelets and growth factors allowing their sustained release.
This scaffolding (fibrin matrix) localizes the growth factors increasing their concentration at the desired location and guiding tissue regeneration. Studies have shown that platelet growth factors are released for up to seven days, supporting collagen production and increased vascularization, which results in overall skin improvements.